单选题 适中0.65 引用7 组卷520
25℃时,用0.0500 mol·L-1H2C2O4溶液滴定 25.00 mL0.l000mol·L-1NaOH溶液所得滴定曲线如图。下列说法正确的是
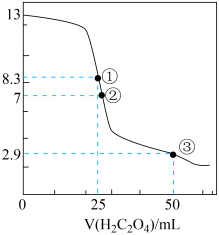

| A.点①所示溶液中:c(H+)+c(H2C2O4)+c(HC2O4-)=c(OH-) |
| B.点②所示溶液中:c(HC2O4-)+c(C2O42-)=c(Na+) |
| C.点③所示溶液中:c(Na+)>c(HC2O4-)>c(H2C2O4)>c(C2O42-) |
| D.滴定过程中可能出现:c(Na+)>c(C2O42-)=c(HC2O4-)>c(H+)>c(OH-) |
2017·江苏南通·二模
类题推荐
常温下,用0.10mol·L-1KOH溶液滴定10.00mL0.10mol·L-1H2C2O4(二元弱酸)溶,所得滴定曲线如图所示(混合溶液的体积可看成混合前溶液的体积之和)。下列说法正确的是( )
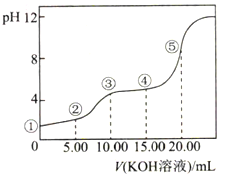

| A.点①所示溶液中:c(H+)/c(OH-)=1012 |
| B.点②所示溶液中:c(K+)+c(H+)=c(HC2O4-)+c(C2O42-)+c(OH-) |
| C.点③所示溶液中:c(K+)>c(HC2O4-)>c(H2C2O4)>c(C2O42-) |
| D.点④所示溶液中:c(K+)+c(H2C2O4)+c(HC2O4-)+ c(C2O42-)=0.10mol·L-1 |
常温下,用0.10mol·L-1KOH溶液滴定10.00mL0.10mol·L-1H2C2O4(二元弱酸)溶,所得滴定曲线如图所示(混合溶液的体积可看成混合前溶液的体积之和)。下列说法正确的是( )


| A.点①所示溶液中:c(H+)/c(OH-)=1012 |
| B.点②所示溶液中:c(K+)+c(H+)=c(HC2O4-)+c(C2O42-)+c(OH-) |
| C.点③所示溶液中:c(K+)>c(HC2O4-)>c(H2C2O4)>c(C2O42-) |
| D.点④所示溶液中:c(K+)+c(H2C2O4)+c(HC2O4-)+ c(C2O42-)=0.10mol·L-1 |
用0.1mol·L-1的NaOH溶液滴定25mL0.1mol·L-1的H2C2O4(草酸)溶液的滴定曲线如图所示。下列说法不正确的是
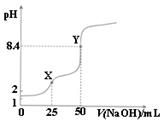

| A.草酸是弱酸 |
| B.X点:c(H2C2O4)+c(HC2O4-)=c(Na+)-c(C2O42-) |
| C.Y点:c(H2C2O4)+c(HC2O4-)=c(OH-)-c(H+) |
| D.滴定过程中始终存在:c(OH-)+2c(C2O42-)+ c(HC2O4-)= c(Na+)+ c(H+) |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


