单选题 适中0.64 引用1 组卷164
已知氢气的燃烧热为286 kJ/mol;且H2O(g) = H2O(l) ΔH = —44 kJ/mol。下列热化学方程式正确的是
| A.2H2(g) + O2(g) = 2H2O(g)ΔH = —286 kJ/mol |
| B.2H2(g) + O2(g) = 2H2O(g)ΔH = —572 kJ/mol |
C.H2(g) + 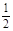 O2(g) = H2O(g)ΔH = —330 kJ/mol O2(g) = H2O(g)ΔH = —330 kJ/mol |
D.H2O(g) = H2(g) + 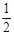 O2(g)ΔH=+242kJ/mol O2(g)ΔH=+242kJ/mol |
14-15高二下·北京房山·期中
类题推荐
下列热化学方程式书写正确的是
| A.2SO2+O2 |
| B.H2(g)+1/2O2(g)= H2O(l) ΔH =-285.8 kJ·molˉl |
| C.2H2 (g) + O2 (g)= 2H2O(l) ΔH =+571.6 kJ·molˉl |
| D.C(s)+ O2(g)= CO2(g) ΔH =+393.5 kJ·molˉl |
已知H—H键的键能为436 kJ/mol,O=O键的键能为498 kJ/mol,根据热化学方程式H2(g)+ O2(g)=H2O(l) ΔH=-286 kJ/mol,判断H2O分子中O—H键的键能为
O2(g)=H2O(l) ΔH=-286 kJ/mol,判断H2O分子中O—H键的键能为
| A.485.5 kJ/mol | B.610 kJ/mol |
| C.917 kJ/mol | D.1 220 kJ/mol |
已知H—H键的键能为436 kJ/mol,O=O键的键能为498 kJ/mol,根据热化学方程式H2(g)+ O2(g)=H2O(l) ΔH=-286 kJ/mol,判断H2O分子中O—H键的键能为
O2(g)=H2O(l) ΔH=-286 kJ/mol,判断H2O分子中O—H键的键能为
| A.485.5 kJ/mol | B.610 kJ/mol |
| C.917 kJ/mol | D.1 220 kJ/mol |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


