加氢是对温室气体的有效转化,也是生成再生能源与化工原料重要途径。
(1)由

加氢生成

的反应为

①若

为标准摩尔生成焓,其定义为标准状态下,由稳定相态的单质生成1mol该物质的焓变。则该反应的反应热
 ___________
___________
②若在一定温度下的容积固定的密闭容器中进行该反应,则可以提高

平衡转化率的措施有
___________(写出两种)。
③下列说法可以证明该反应已达到平衡状态的是
___________(填字母)。
a.

b.

的物质的量分数不再改变
c.容器内气体密度不再改变
d.

和

的浓度之比为1:2
e.混合气的平均摩尔质量不再变化
f.单位时间内生成

的同时又消耗

④

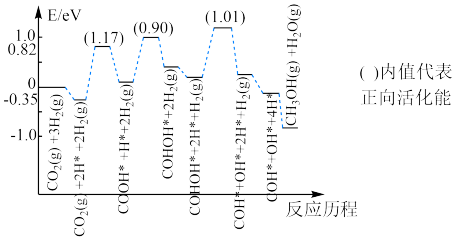
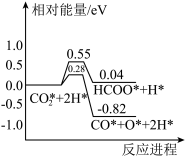
催化加氢合成

过程中,

活化的可能途径如有图所示,CO是

活化的优势中间体,原因是
___________。
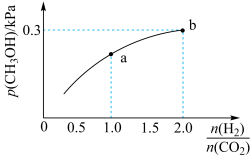
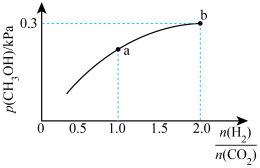
(2)一定条件下

与

也可以生成

,某温度下在容积为2L的恒容密闭容器中充入

和一定量

发生反应:

。

的平衡分压与起始投料比

的变化关系如图所示,已知b点对应投料比的起始压强为1.5kPa,则
 ___________
___________。b点时,再充入

和

,使两者分压均增大0.2kPa,则此时平衡
___________(填“正向”、“逆向”或“不”)移动。
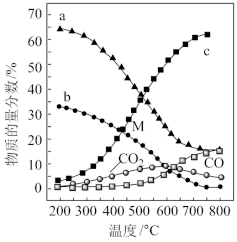
(3)一定条件下

与

还可以生成甲酸,方程式为


。恒定压强为100kPa时,向密闭容器中充入一定量的HCOOH气体,

除了能分解成

和

,还能分解为CO和

,反应为:


,则此平衡体系中CO或

的选择性和HCOOH的转化率随温度变化曲线如图所示。

提示:CO(或氢气)的选择性

图中随着温度升高,HCOOH的转化率增大,请解释CO的选择性下降可能的原因:
___________;400°C时

的体积分数为
___________。