单选题 适中0.65 引用1 组卷173
化学电源在日常生活和高科技领域中都有广泛应用。下列说法不正确的是
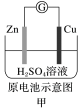


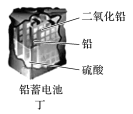




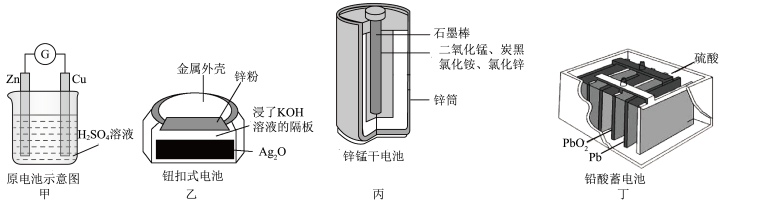
| A.甲:Zn2+向Cu电极方向移动,溶液中H+浓度减小 |
| B.乙:正极的电极反应式为Zn-2e-+2OH-=Zn(OH)2 |
| C.丙:锌筒作负极,发生氧化反应,锌筒会变薄 |
| D.丁:放电一段时间后,电解质溶液的浓度减小,两极质量增加 |
20-21高一下·湖北恩施·阶段练习
类题推荐
化学电源在日常生活和高科技领域中都有广泛应用。下列说法正确的是
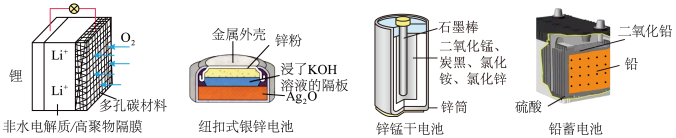

| A.图甲:锂电池放电时,电解质溶液中Li+向锂电极迁移 |
| B.图乙:负极的电极反应式为 |
| C.图丙:锌筒作负极,发生氧化反应,锌筒会变薄 |
| D.图丁:电池放电过程中,硫酸浓度不断减小 |
化学电源在日常生活和高科技领域中都有广泛应用,下列说法不正确的是


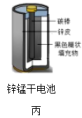





| A.甲图中Zn为负极,Cu电极发生还原反应 |
| B.乙图中正极的电极反应式为: |
| C.丙图中锌筒作负极,发生氧化反应,锌筒会变薄 |
| D.丁图中使用一段时间后,电解质溶液的酸性减弱,导电能力下降 |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网