单选题 适中0.65 引用1 组卷124
常温下,向 溶液中逐滴加入
溶液中逐滴加入 氨水,所得溶液
氨水,所得溶液 及导电能力变化如图所示。下列分析正确的是
及导电能力变化如图所示。下列分析正确的是



| A. |
| B.常温下, |
| C. |
| D. |
22-23高二上·吉林通化·阶段练习
类题推荐
常温下,向10mL0.1mol·L-1CuCl2溶液中滴入0.1mol·L-l的Na2S溶液,溶液中-lgc(Cu2+)随滴入的Na2S溶液体积的变化如图所示。下列叙述正确的是
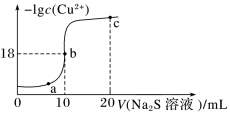

| A.Na2S溶液中:c(S2-)+c(HS-)+c(H2S)=2c(Na+) |
| B.Ksp(CuS)的数量级为10-36 |
| C.a、b、c三点对应溶液中,b点水的电离程度最大 |
| D.c点对应溶液成分只有Na2S |
常温下向10mL0.1mol/L的HR溶液中逐滴加入0.1mol/L的氨水,所得溶液pH及导电性变化如图。下列分析正确的是
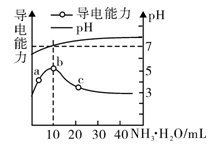

| A.a~c点,溶液中离子数目先增大后减小 |
| B.b点溶液pH=7说明c(NH4+)=c(R-) |
| C.c点溶液存在c(NH4+)>c(R-)>c(H+)>c(OH-) |
| D.b、c两点对应溶液中,水的电离程度相同 |
常温下,向10mL0.1mol/L的某酸(HA) 溶液中逐滴加入0.1mol/L的氨水,所得溶液的pH及导电能力与加入氨水的体积关系如图所示。下列说法中不正确的是
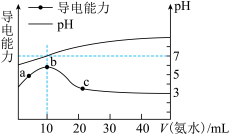

| A.HA为弱酸 |
| B.b点对应溶液pH=7,此时HA溶液与氨水恰好完全反应 |
| C.c点对应的溶液存在c(OH-)=c(H+)+c(HA) |
| D.a、b、c三点中,b点对应的溶液中水的电离程度最大 |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


