单选题 较易0.85 引用9 组卷287
普通锌锰干电池的构造如图所示,其电池反应的方程式为: ,下列说法
,下列说法正确 的是
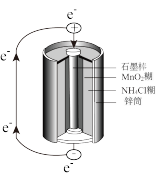

| A.石墨作负极 |
| B.锌发生还原反应 |
| C.电池工作时,电子从石墨经导线流向锌 |
| D.正极的电极反应为: |
20-21高一下·全国·课后作业
类题推荐
纽扣式银锌电池的构造如图所示,电池的总反应为:Zn+Ag2O+H2O=Zn(OH)2+2Ag。下列说法不正确的是
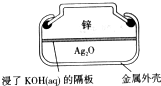

| A.Zn作负极 | B.电子经外电路流向Ag2O极 |
| C.K+向Zn极移动 | D.正极反应:Ag2O+2H2O+2e-=2Ag+2OH- |
关于右图所示的原电池,下列说法正确的是
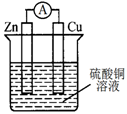

| A.电子从锌电极通过电流表流向铜电极 |
| B.锌电极发生还原反应,铜电极发生氧化反应 |
| C.反应后,锌电极的质量减小 |
| D.铜电极作正极,发生的电极反应为2H++2e-═H2↑ |
纽扣式银锌电池的构造如图所示,电池的总反应为:Zn+Ag2O+H2O=Zn(OH)2+2Ag,下列说法不正确的是
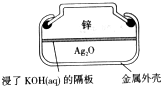

| A.Zn作负极 |
| B.电子经外电路流向Ag2O极 |
| C.K+向Zn极移动 |
| D.正极反应:Ag2O+H2O+2e-=2Ag+2OH- |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


