单选题 适中0.65 引用1 组卷92
用甲醚 燃料电池,电解
燃料电池,电解 溶液制取NaOH溶液和硫酸的装置如下图所示。下列说法正确的是
溶液制取NaOH溶液和硫酸的装置如下图所示。下列说法正确的是
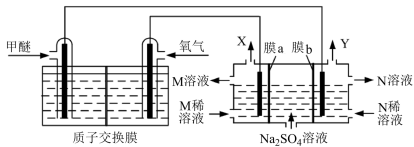

| A.M为NaOH、N为硫酸 |
| B.膜a、膜b依次为阳离子交换膜和阴离子交换膜 |
| C.燃料电池的负极反应式为 |
| D.反应的甲醚与生成气体Y的物质的量之比为1∶3 |
21-22高二上·山西晋城·阶段练习
类题推荐
利用CH4燃料电池电解制备Ca(H2PO4)2,装置如图所示。下列说法正确的是
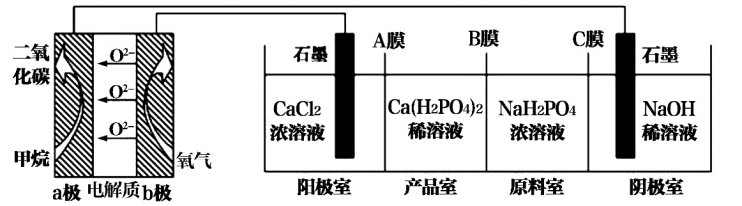

| A.该装置能得到副产物NaOH、H2、Cl2 |
| B.A膜和C膜均为阴离子交换膜,B为阳离子交换膜 |
| C.a极的电极反应式:CH4+2H2O-8e-=CO2+8H+ |
| D.理论上,b极上通入标况下2.24LO2,产品室增加0.2mol Ca(H2PO4)2 |
利用CH4燃料电池电解制备Ca(H2PO4)并得到副产物NaOH、H2、Cl2,装置如图所示。下列说法不正确的是
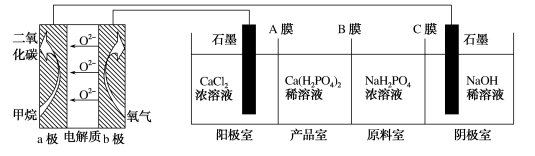

| A.a极反应: |
| B.A膜和C膜均为阳离子交换膜 |
| C.可用铁电极替换阴极的石墨电极 |
| D.a极上通入2.24L甲烷,阳极室Ca2+减少0.4mol |
利用CH4燃料电池电解制备Ca(H2PO4)2并得到副产物NaOH、H2、Cl2,装置如图所示。下列说法正确的是
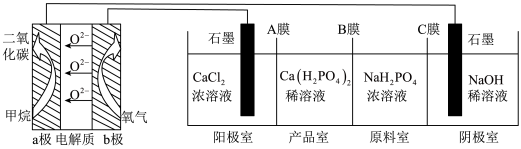

| A.a极反应:CH4+8e-+4O2-=CO2+2H2O |
| B.A膜和C膜均为阴离子交换膜 |
| C.可用铁电极替换阴极的石墨电极 |
| D.a极上通入2.24 L甲烷,阳极室Ca2+减少0.4 mol |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


