单选题 较易0.85 引用1 组卷130
已知热化学方程式:
①CO(g)+1/2O2(g)=CO2(g) ΔΗ= -283.0kJ/mol
②H2(g)+1/2O2(g)=H2O(g) ΔΗ= -241.8kJ/mol
据此判断下列说法正确的是
①CO(g)+1/2O2(g)=CO2(g) ΔΗ= -283.0kJ/mol
②H2(g)+1/2O2(g)=H2O(g) ΔΗ= -241.8kJ/mol
据此判断下列说法正确的是
| A.H2(g)+1/2O2(g)=H2O(l) ΔΗ>-241.8kJ/mol |
| B.H2(g)的燃烧热ΔΗ =-241.8 kJ/mol |
| C.H2(g)转变成H2O(g)的化学反应一定放出能量 |
| D.CO(g)+ H2O(g)= CO2(g)+ H2(g)的ΔΗ =-41.2kJ/mol |
21-22高二上·黑龙江鸡西·期中
类题推荐
已知热化学方程式:①CO(g)+1/2O2(g)=CO2(g) ΔH=-283.0kJ·mol-1
②H2(g)+1/2O2(g)=H2O(g) ΔH=-241.8kJ·mol-1
则下列说法正确的是

②H2(g)+1/2O2(g)=H2O(g) ΔH=-241.8kJ·mol-1
则下列说法正确的是

| A.H2的燃烧热为241.8kJ·mol-1 |
| B.由反应①、②可知上图所示的热化学方程式为CO(g)+H2O(g)=CO2(g)+H2(g) ΔH=-41.2kJ·mol-1 |
| C.H2(g)转变成H2O(g)的化学反应一定要吸收能量 |
| D.根据②推知反应H2(g)+1/2O2(g)=H2O(l) ΔH>-241.8kJ·mol-1 |
已知热化学方程式:
①CO(g)+1/2O2(g)=CO2(g)ΔH=-283.0kJ·mol-1
②H2(g)+1/2O2(g)=H2O(g)ΔH=-241.8 kJ·mol-1
则下列说法正确的是( )
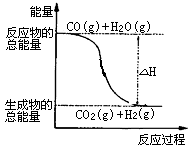
①CO(g)+1/2O2(g)=CO2(g)ΔH=-283.0kJ·mol-1
②H2(g)+1/2O2(g)=H2O(g)ΔH=-241.8 kJ·mol-1
则下列说法正确的是( )

| A.H2的燃烧热为241.8kJ·mol-1 |
| B.由反应①、②可知上图所示的热化学方程式为CO(g)+H2O(g)=CO2(g)+H2(g)ΔH=-41.2 kJ·mol-1 |
| C.H2(g)转变成H2O(g)的化学反应一定要放出能量 |
| D.根据②推知反应H2(g)+1/2O2(g)=H2O(l)ΔH>-241.8 kJ·mol-1 |
燃烧热是指在25℃,101kPa,1mol可燃物完全燃烧生成稳定的化合物时放出的热量。已知热化学方程式:①CO(g)+1/2 O2(g)=CO2(g) ΔH= -283.0kJ·mol-1
②H2(g)+1/2O2(g)=H2O(g) ΔH= -241.8kJ·mol-1
则下列说法正确的是

②H2(g)+1/2O2(g)=H2O(g) ΔH= -241.8kJ·mol-1
则下列说法正确的是

| A.H2的燃烧热为241.8 kJ·mol-1 |
B.由反应①、②可知如图所示的热化学方程式为CO(g)+H2O(g)  CO2(g)+H2(g) ΔH= -41.2kJ·mol-1 CO2(g)+H2(g) ΔH= -41.2kJ·mol-1 |
| C.H2(g)转变成H2O(g)的化学反应一定要释放能量 |
| D.根据②推知反应H2(g)+1/2O2(g)=H2O(l) ΔH> -241.8 kJ·mol-1 |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


