单选题 较难0.4 引用6 组卷1439
常温下,用 溶液分别滴定
溶液分别滴定 浓度均为
浓度均为 的
的 溶液和
溶液和 溶液,滴定曲线如图。下列说法正确的是
溶液,滴定曲线如图。下列说法正确的是


| A.电离平衡常数: |
| B.水的电离程度:④>①>③>② |
| C.点①和点②所示溶液中: |
| D.点③和点④所示溶液中均有: |
2021·重庆沙坪坝·模拟预测
类题推荐
常温下,用0.10 mol·L-1NaOH溶液分别滴定20.00 mL浓度均为0.10 mol·L-1CH3COOH溶液和HCN溶液,所得滴定曲线如图。下列说法不正确 的是
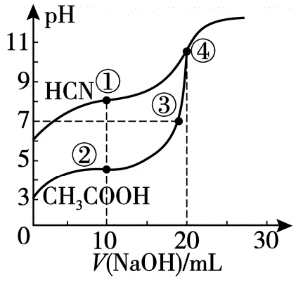

| A.点①所示溶液中,CN-的水解程度大于HCN的电离程度 |
| B.点①和点②所示溶液中:c(CN-)>c(CH3COO-) |
| C.点③所示溶液中:c(Na+)=c(CH3COO-)>c(H+)=c(OH-) |
| D.点④所示溶液中:c(Na+)>c(CH3COO-)>c(OH-)>c(H+) |
室温下,用0.10 mol/L NaOH溶液分别滴定20.00 mL浓度均为0.10 mol/L的CH3COOH 溶液和HCN溶液所得滴定曲线如图所示。下列说法正确的是
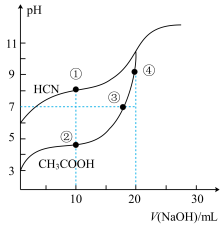

| A.室温下,电离平衡常数:K(HCN)>K(CH3COOH) |
| B.点①和点②所示溶液中:c(CH3COO-)>c(CN-) |
| C.点③所示的溶液中:c(CH3COO-)+c(OH-)=c(CH3COOH)+c(H+) |
| D.点④所示的溶液中:c(Na+)>c(OH-)>c(CH3COO-)>c(H+) |
常温下,用0.100mol•L-1NaOH溶液分别滴定20.00mL浓度均0.100mol•L-1CH3COOH溶液和HCN溶液,所得滴定曲线如图。下列说法不正确的是( )
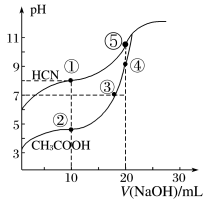

| A.点①和点②所示溶液中:c(CH3COO-)>c(CN-) |
| B.点③时:c(Na+)=c(CH3COO-)>c(H+)=c(OH-) |
| C.点③和点④所示溶液中水的电离程度:③<④ |
| D.点④溶液中阴离子总数小于点⑤ |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


