单选题 较易0.85 引用1 组卷278
下列选项中的图象与其解释或得出的结论对应关系正确的是
| A | B | C | D | |
| 图象 | 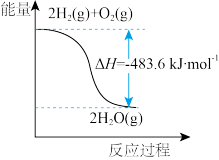 | 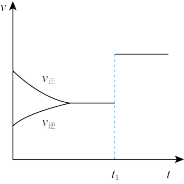 | 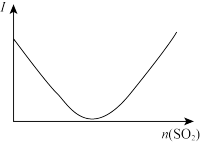 | 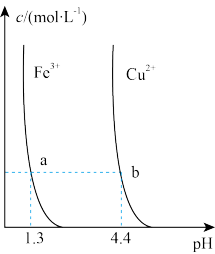 |
| 解释或结论 | 热化学方程式是 | 某恒容容器中发生反应:X(g)+Y(g) | 向NaOH溶液中通入 | T℃时, |
| A.A | B.B | C.C | D.D |
18-19高二上·河南焦作·期末
类题推荐
下列关于各图象的解释或结论正确的是( )
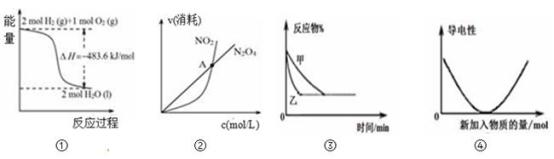

| A.图①可知:热化学方程式是H2(g)+1/2O2(g)=H2O(g)△H=﹣241.8kJ/mol |
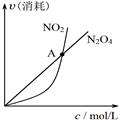
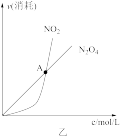
B.图②可知:对于恒温恒容条件下的反应2NO2(g) N2O4(g),A点为平衡状态 N2O4(g),A点为平衡状态 |
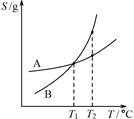
C.图③可表示:压强对2A(g)+2B(g) 3C(g)+D(s)的影响 3C(g)+D(s)的影响 |
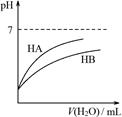
| D.图④可表示:Ba(OH)2溶液中滴加稀硫酸,溶液导电性的变化 |
下列关于各图象的解释或结论正确的是

甲 乙 丙 丁




甲 乙 丙 丁
A.由甲可知:热化学方程式是H2(g) + O2(g) = H2O(g) △H=﹣241.8kJ/mol O2(g) = H2O(g) △H=﹣241.8kJ/mol |
| B.由乙可知:对于恒温恒容条件下的反应2NO2(g) ⇌N2O4(g),A点为平衡状态 |
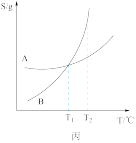
| C.由丙可知:将A、B饱和溶液分别由T1℃升温至T2℃时,溶质的质量分数B>A |
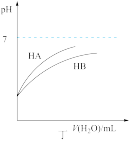
| D.由丁可知:同温度、同浓度的NaA溶液与NaB溶液相比,其pH前者小于后者 |
下列关于各图象的解释或结论错误的是
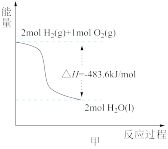




| A.由甲可知:热化学方程式是H2(g)+1/2O2(g)=H2O(l) ∆H =-241.8kJ·mol-1 |
| B.由乙可知:对于恒温恒容条件下的反应2NO2(g) ⇌N2O4(g),A点为平衡状态 |
| C.由丙可知:将A、B饱和溶液分别由T1℃升温至T2℃时,溶质的质量分数A=B |
| D.由丁可知:同温度、同浓度的NaA溶液与NaB溶液相比,其pH前者小于后者 |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网


