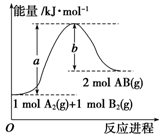
单选题 较易0.85 引用5 组卷171
化学反应N2(g)+3H2(g)===2NH3(l)的能量变化如图所示,则该反应的ΔH等于 ( )
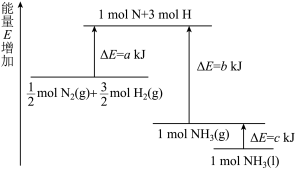

| A.2(a-b-c) kJ·mol-1 | B.-2(b-a) kJ·mol-1 |
| C.- (b+c-a) kJ·mol-1 | D.(a+b) kJ·mol -1 -1 |
18-19高一下·内蒙古通辽·期末
类题推荐
已知H—H、H—O和O O键的键能分别为436 kJ·mol-1、463 kJ·mol-1和495 kJ·mol-1,下列热化学方程式正确的是
O键的键能分别为436 kJ·mol-1、463 kJ·mol-1和495 kJ·mol-1,下列热化学方程式正确的是
 O键的键能分别为436 kJ·mol-1、463 kJ·mol-1和495 kJ·mol-1,下列热化学方程式正确的是
O键的键能分别为436 kJ·mol-1、463 kJ·mol-1和495 kJ·mol-1,下列热化学方程式正确的是A.H2O(g) H2(g)+ H2(g)+ |
B.H2O(g) H2(g)+ H2(g)+ |
C.2H2(g)+O2(g) 2H2O(g) ΔH=+485 kJ·mol-1 2H2O(g) ΔH=+485 kJ·mol-1 |
D.2H2(g)+O2(g) 2H2O(g) ΔH=-485 kJ·mol-1 2H2O(g) ΔH=-485 kJ·mol-1 |
已知化学反应A2(g)+B2(g)===2AB(s)的能量变化如图所示,判断下列叙述中错误的是
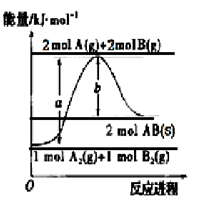

| A.该反应热ΔH=+(a-b)kJ·mol-1 |
| B.AB(s)=== A(g)+B(g) ΔH=+b/2kJ·mol-1 |
| C.断裂1 mol A-A键和1 mol B-B键,吸收a kJ能量 |
| D.该反应在任何条件下都不可能自发进行 |
组卷网是一个信息分享及获取的平台,不能确保所有知识产权权属清晰,如您发现相关试题侵犯您的合法权益,请联系组卷网